 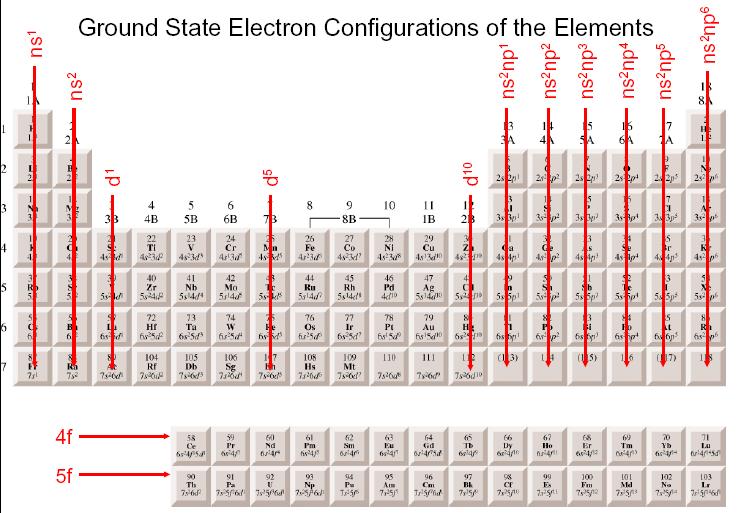
So here we have the visual representation of the atom or using the electron configuration to justify this representation of the chlorine atom. So we have one, 2345 six, and then seven. So let's add the additional two electrons that we need. And then we can say here that the outer shell has seven electrons total in it. The second shell, and equals two has a total of eight electrons in it. If we have a representation of the atom like we do here, the first shell has two electrons in it. So here this will be shell one shell to and shell three. Remember, the number in front tells us the shell number. It's electron configuration is one s to to us to two p 63 s 23 p five. And we're going to say here that your atomic number can be thought of as your total number of electrons and that equals just all your valence electrons, plus your inner core electrons. The inner core electrons are just all the remaining electrons that are not valence electrons. When we say valence electrons, the valence electrons are just your outer shell electrons involved in forming chemical bonds. So basically not the transition metals were going to say the number of Valence electrons equals the group number of the element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
